Droplet microfluidics as a powerful tool for high-throughput assays in biology and chemistry
 Droplet microfluidics is indeed one of the most successful domains of microfluidics both at academic and industrial levels. Droplet-based microfluidics is the technology that deals with the formation and manipulation of uniform and micron-sized droplets at extremely high rates. What makes droplet microfluidics unique is the very high production rate of the droplets such that it enables thousands of assays to be done in parallel. Additionally, droplet microfluidics brings about a much higher analysis resolution in comparison to the conventional techniques. The droplet sizes are adjustable and can be reduced to sizes comparable to the sizes of individual cells. The small confinement allows the individual cells to be trapped within the droplets thus enabling high-throughput single-cell analysis. Droplet generator chips are quite robust and can run for long hours with minimal supervision. The droplets are discrete which eliminates the risk of cross-contamination. The applications of droplet microfluidics are very broad and range from microparticle synthesis to molecular biology and microorganism studies.
Droplet microfluidics is indeed one of the most successful domains of microfluidics both at academic and industrial levels. Droplet-based microfluidics is the technology that deals with the formation and manipulation of uniform and micron-sized droplets at extremely high rates. What makes droplet microfluidics unique is the very high production rate of the droplets such that it enables thousands of assays to be done in parallel. Additionally, droplet microfluidics brings about a much higher analysis resolution in comparison to the conventional techniques. The droplet sizes are adjustable and can be reduced to sizes comparable to the sizes of individual cells. The small confinement allows the individual cells to be trapped within the droplets thus enabling high-throughput single-cell analysis. Droplet generator chips are quite robust and can run for long hours with minimal supervision. The droplets are discrete which eliminates the risk of cross-contamination. The applications of droplet microfluidics are very broad and range from microparticle synthesis to molecular biology and microorganism studies.
How does droplet microfluidic work?
So, how do microfluidic droplet generator chips work? How are they capable of making such small and uniform droplets at this unprecedented rate? It all related to the capacity of droplet-based microfluidic devices for making small nozzles. Flow-focusing and T-channel droplet microchips are two of the most common nozzle designs.
 In flow-focusing microfluidic chips, the dispersed phase microchannel and the carrier microchannel form an intersection. The carrier phase meets the dispersed phase from the sides and at a higher flow rate. The carrier phase pushes the dispersed phase from the size causing it to burst into droplets. In T-channel chips, the dispersed phase t-bones the carrier microchannel. The carrier phase shears the dispersed phase to break them into droplets. The size of the droplets and frequency of droplet generation is highly dependent on the ratio of the dispersed phase and continuous phase flow rates. There is a flow-rate threshold that allows the generation of uniform droplets. Above or below this threshold will result in polydisperse droplets or no droplets at all. Care should be taken to analyze the droplet generator before use to determine the flow-rates ratios at which droplets are uniform. The reagents can also affect the droplet size and uniformity. In other words, a flow rate-ratio that led to stable droplets for a particular chemical does not necessarily result in uniform droplets of the same size if the chemicals change. The specification sheet used for a particular chip cannot be used for another chip with different microfluidic channel dimensions. However, the values that were working for another chip with comparable dimensions can give a good idea of a starting point for characterizing the other chips.
In flow-focusing microfluidic chips, the dispersed phase microchannel and the carrier microchannel form an intersection. The carrier phase meets the dispersed phase from the sides and at a higher flow rate. The carrier phase pushes the dispersed phase from the size causing it to burst into droplets. In T-channel chips, the dispersed phase t-bones the carrier microchannel. The carrier phase shears the dispersed phase to break them into droplets. The size of the droplets and frequency of droplet generation is highly dependent on the ratio of the dispersed phase and continuous phase flow rates. There is a flow-rate threshold that allows the generation of uniform droplets. Above or below this threshold will result in polydisperse droplets or no droplets at all. Care should be taken to analyze the droplet generator before use to determine the flow-rates ratios at which droplets are uniform. The reagents can also affect the droplet size and uniformity. In other words, a flow rate-ratio that led to stable droplets for a particular chemical does not necessarily result in uniform droplets of the same size if the chemicals change. The specification sheet used for a particular chip cannot be used for another chip with different microfluidic channel dimensions. However, the values that were working for another chip with comparable dimensions can give a good idea of a starting point for characterizing the other chips.
Are droplet microfluidics and digital microfluidics the same thing?
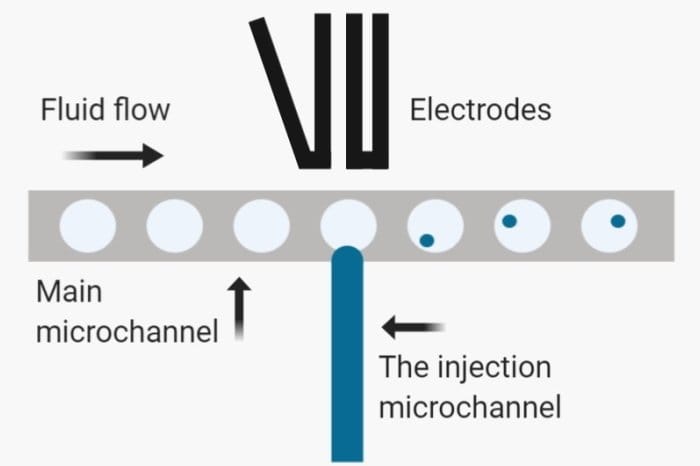
It is of note that droplet microfluidics should not be confused with digital microfluidics. Digital microfluidics is the science and technology of moving and manipulating small droplets on a platform that consists of insulated electrodes. Turning the electrodes on-off sequentially can move the droplet on the platform. In other words, digital microfluidics deals with discrete droplets and a limited number of droplets. But, droplet microfluidics devices are fabricated and designed to handles large scale production of droplets in a continuous manner.
What are some popular droplet generator designs and where can I purchase them?
 It depends on what your application is. Based on the specifications of your experiment, you might be able to benefit from some of the popular microfluidic droplet generators. For example, for molecular biology and single-cell analysis, Dropseq and inDrop are popular among researchers. Dropseq, developed by McCaroll’s lab at Harvard University, and first introduced in 2015, is a microfluidic droplet generator capable of isolating single cells in 125 µm droplets. uFluidix is endorsed by McCaroll’s Lab as a supplier of dropseq chips. Variations of these chips, such as DroNc-seq, with different dimensions or components, have also been developed to accommodate the needs of similar experiments with slight differences.
It depends on what your application is. Based on the specifications of your experiment, you might be able to benefit from some of the popular microfluidic droplet generators. For example, for molecular biology and single-cell analysis, Dropseq and inDrop are popular among researchers. Dropseq, developed by McCaroll’s lab at Harvard University, and first introduced in 2015, is a microfluidic droplet generator capable of isolating single cells in 125 µm droplets. uFluidix is endorsed by McCaroll’s Lab as a supplier of dropseq chips. Variations of these chips, such as DroNc-seq, with different dimensions or components, have also been developed to accommodate the needs of similar experiments with slight differences.
Read more about what needs to be considered before designing a droplet microfluidic chip
There are many microfluidics methods to manipulate droplets after generation. Learn how droplets can be modified.
Droplet microfluidics is one of the most popular microfluidic domains. Learn about its applications.