
01 Mar Small scale, big results: The advantages of microfabricated devices for protein crystallization
Protein crystallization is a crucial step in determining the structure of proteins, which is essential for understanding their function and developing new drugs. However, the process of crystallization is notoriously difficult and can take a long time to achieve success. In recent years, researchers have been exploring new methods and technologies to improve the efficiency and success rate of protein crystallization. Microfluidics has emerged as a powerful tool for protein crystallization studies. By controlling the microscale environment in which proteins and precipitants interact, microfluidic devices offer advantages over traditional bulk methods, such as improved mixing, enhanced mass transfer, and reduced sample consumption. Microfluidic chip fabrication and microfabrication techniques have enabled the design and production of highly precise and reproducible microfluidic devices for a wide range of applications, including protein crystallization. In this week’s research highlight, we feature an article by MIT researchers that introduces a novel approach for protein crystallization using a droplet microfluidic platform. The platform utilizes a combination of a microfluidic mixer and a droplet generator to generate a population of identical and independent droplets.
“We use a microbatch crystallization setup to show that the range of successful crystallization conditions is expanded by the presence of functionalized nanoparticles. Furthermore, we use a custom machine learning-enabled emulsion crystallization setup to rigorously quantify nucleation parameters. We show that bioconjugate-functionalized nanoparticles can result in up to a 7-fold decrease in the induction time and a 3-fold increase in the nucleation rate of model proteins compared to those in control environments. “, the authors explained.
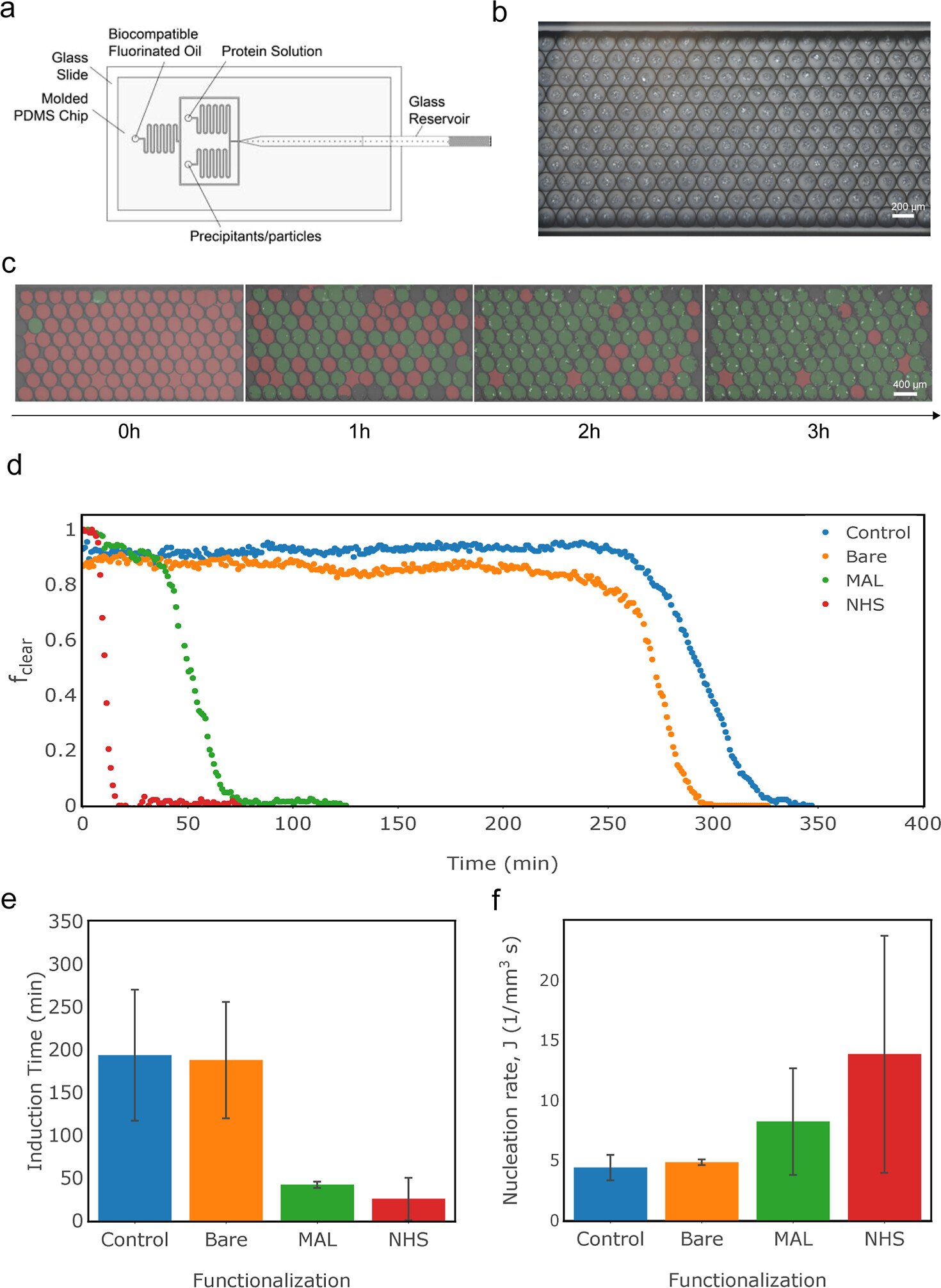
“Lysozyme crystallization; (a) schematic of the microfluidic droplet generator, (b) raw optical image of lysozyme crystals within emulsion droplets at the end of a nucleation experiment, (c) chronophotography images with red/green overlay to indicate droplets containing crystals, (d) fraction of clear droplets as a function of time for each condition (plot shows data from one experiment), (e) induction time, and (f) nucleation rates for lysozyme crystallization as a function of nanoparticle functionalization (bar graphs show average of three sets of droplets; error bars show standard deviation).” Reproduced from Caroline McCue, Henri-Louis Girard, and Kripa K. Varanasi ACS Applied Materials & Interfaces Article ASAP DOI: 10.1021/acsami.2c17208 under Creative Commons Attribution 4.0 International (CC BY 4.0).
The study conducted three sets of experiments to evaluate the effectiveness of their crystallization strategy: vapor diffusion, batch crystallization, and droplet crystallization. Vapor diffusion is a widely used technique to screen crystallization conditions. In this experiment, a sitting or hanging drop of protein solution and precipitants is sealed within an environment containing a reservoir solution with a larger volume and higher precipitant concentration. The transfer of water from the drop to the reservoir as the solution equilibrates drives protein saturation within the drop and initiates nucleation. The researchers used hen egg white lysozyme and sodium chloride in a sodium acetate buffer at pH 4.5. For each experimental condition, 16 μL of nanoparticles were added to 1 mL of solution. The droplets were imaged with an optical microscope after 20 hours at room temperature to screen the crystallization outcomes for crystal size, quality, and quantity.
Batch crystallization was used to measure protein crystallization probability across a range of protein and salt concentrations. Bulk protein solution was mixed with precipitants and then sealed to prevent evaporation. The samples were kept in sealed PCR tubes for the duration of the crystallization phase. Aliquots from these reaction tubes were taken for visualization and imaging at the end in the form of droplets. The researchers used a high lysozyme concentration and a high sodium chloride concentration solution to prepare the protein and salt solutions. Functionalized nanoparticles were then added to achieve the target concentrations and supersaturation levels in each condition. After 72 hours at room temperature, a 30 μL droplet was withdrawn from the bottom of each tube, placed on a microscope slide, and imaged with an optical microscope. The percent of trials that displayed crystals was calculated and analyzed using bootstrap analysis to determine 95% confidence intervals.
Droplet crystallization was used to measure the nucleation rate and induction times of crystallization. The researchers developed a microfluidic platform combining a microfluidic mixer and an emulsion generator to generate a population of identical, independent droplets. A concentration of lysozyme or insulin was used in the protein stream, and NaCl in sodium acetate buffer containing the nanoparticles was used as the precipitant stream. These streams were mixed within the microfluidic chip for a final concentration of protein and NaCl in sodium acetate buffer within the emulsions. The droplets were imaged at regular time intervals using a microscope connected to a camera with polarizers installed at right angles to each other along the light path before and after the capillary. The researchers analyzed the fraction of clear drops at each time point to determine the induction time and the nucleation rate of crystallization.
“Bioconjugate functionalized nanoparticles could even be integrated into existing microfluidic devices for separation and purification. Future studies are needed to examine the effects of these nanoparticles on crystallization of proteins in mixed solutions and to implement the use of such nanoparticle seeds into current crystallizer workflows. The use of these bioconjugate-functionalized nanoparticles could enable protein crystallization as a purification method and lower the cost of downstream processing, which could lead to a reduction in the cost of crucial biologic drugs. “, the authors concluded.
Figures and the abstract are reproduced from Caroline McCue, Henri-Louis Girard, and Kripa K. Varanasi ACS Applied Materials & Interfaces Article ASAP DOI: 10.1021/acsami.2c17208 under Creative Commons Attribution 4.0 International (CC BY 4.0)
Read the original article: Enhancing Protein Crystal Nucleation Using In Situ Templating on Bioconjugate-Functionalized Nanoparticles and Machine Learning


