17 Mar Droplet microfluidics enables high-throughput CRISPR-based gene editing and screening
Innovations in gene editing are critical for advancing biological research and biotechnology. A recent study published in Microsystems & Nanoengineering presents a groundbreaking method that combines CRISPR technology with droplet microfluidics, significantly advancing the field of genetic engineering.
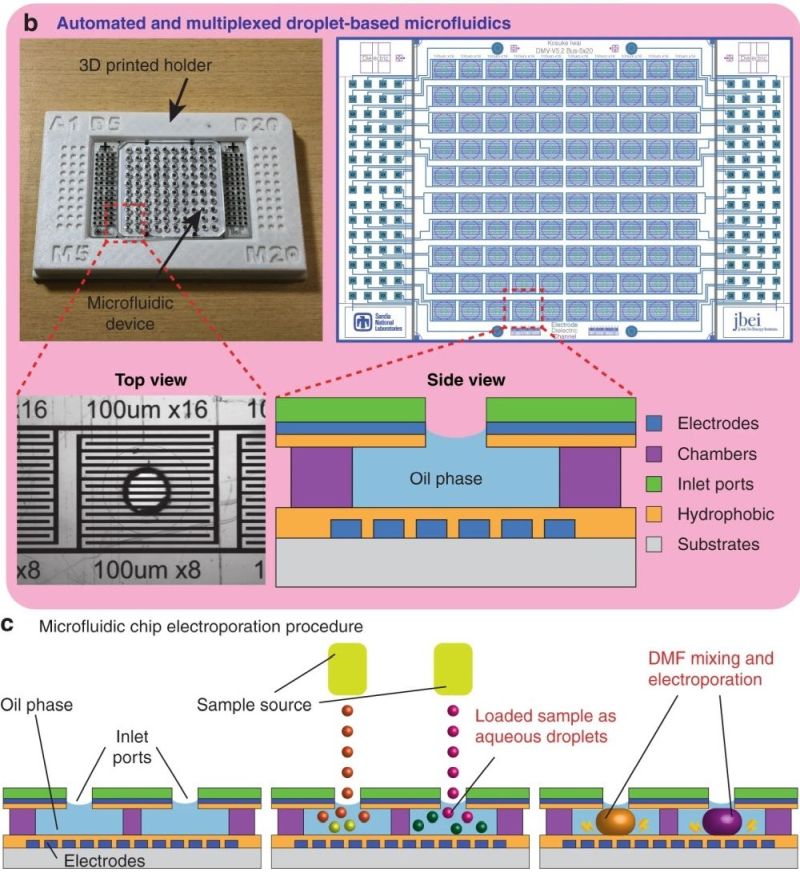
“We present a droplet-based microfluidic system that enables CRISPR-based gene editing and high-throughput screening on a chip. The microfluidic device contains a 10 × 10 element array, and each element contains sets of electrodes for two electric field-actuated operations: electrowetting for merging droplets to mix reagents and electroporation for transformation. This device can perform up to 100 genetic modification reactions in parallel, providing a scalable platform for generating the large number of engineered strains required for the combinatorial optimization of genetic pathways and predictable bioengineering. “, the authors explained.
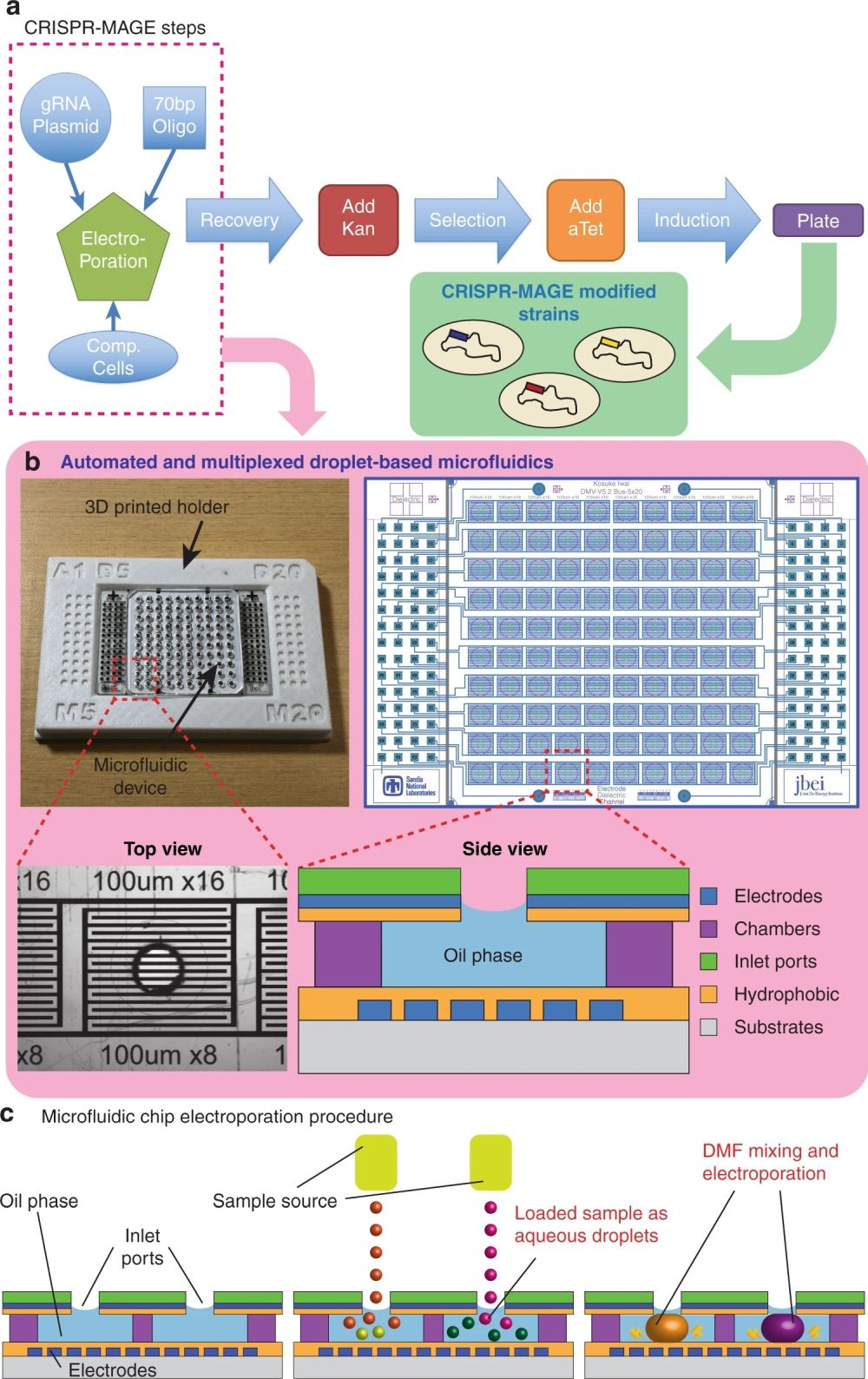
“a CRISPR-MAGE steps. The ones inside the box are performed on the chip. Cells are removed from the chip after the recovery step for induction and plating. b The microfluidic chip in a 3D printed holder (left), the electrode pattern (right), a top-view of an individual well, and a side-view schematic of a well. The chip is designed to contain 100 discrete reaction chambers with individually addressable electrodes for multiplexed CRISPR-MAGE recombineering, and its 384-well format design can be interfaced with lab automation equipment. c Droplets containing plasmids and cells are dispensed into each chamber through the inlet port, mixed by electrowetting, and electroporated by applying a voltage pulse.” Reproduced under Creative Commons Attribution 4.0 International License from Iwai, K., Wehrs, M., Garber, M. et al. Scalable and automated CRISPR-based strain engineering using droplet microfluidics. Microsyst Nanoeng 8, 31 (2022)..
The core of this revolutionary methodology lies in the microfluidic device, engineered to handle up to 100 genetic modification reactions simultaneously. This microfluidic chip, a marvel of microfabrication, features a 10×10 element array, with each element equipped with electrodes for two electric field-actuated operations: electrowetting for merging droplets and electroporation for transformation.
The research showcased the microfluidic device’s capability through CRISPR-based Multiplex Automated Genome Engineering (MAGE), optimizing two E. coli test cases. The researchers demonstrated the microfluidic system’s capability by engineering E. coli strains. The first targeted the enzyme galactokinase (galK) disruption, and the second involved engineering the glutamine synthetase gene (glnA) and blue-pigment synthetase gene (bpsA) for enhanced indigoidine production. The system uses electrowetting for merging droplets, mixing reagents effectively. Electroporation transforms these droplets, making the device apt for comprehensive genetic engineering.
This research marks a significant milestone in the field of genetic engineering, showcasing how microfluidic CRISPR technology can revolutionize our approach to strain engineering and synthetic biology.
“The automated platform for multiplexed transformation holds the promise of accelerating the design-build-test-learn cycle and optimizing biosynthetic pathways. This technology could provide the technological basis for self-driving bioengineering labs, which couples automated experimentation and AI systems that propose experiments and gauge the resulting data to accelerate the bioengineering process. “, the authors concluded.
Figures and the abstract are reproduced from Iwai, K., Wehrs, M., Garber, M. et al. Scalable and automated CRISPR-based strain engineering using droplet microfluidics. Microsyst Nanoeng 8, 31 (2022). https://doi.org/10.1038/s41378-022-00357-3 under Creative Commons Attribution 4.0 International License
Read the original article: Scalable and automated CRISPR-based strain engineering using droplet microfluidics


